|
This gives you a nitrocellulose membrane that is imprinted with the same protein bands as the gel. You want the negative charge to be on the side of the gel and the positive charge to be on the side of the nitrocellulose membrane to drive the negatively charged proteins over to the positively charged nitrocellulose membrane. Place a nitrocellulose membrane on the gel and, using electrophoresis, drive the protein (polypeptide) bands onto the nitrocellulose membrane. Separate the proteins using SDS-polyacrylamide gel electrophoresis (also known as SDS-PAGE). Let's look at this technique in greater detail.ġ. See the section on RIP for more information, as well as a helpful comparative chart that illustrates the differences between these two techniques. Also, if a protein is degraded quickly, Western blotting won't detect it well you'll need to use (RIP). If you are interested in the rate of synthesis of a protein, Radio-Immune Precipitation (RIP) may be the best assay for you. Western blotting tells you how much protein has accumulated in cells. 
You will use this antibody as a probe to detect the protein of interest. 
So you must be able to produce at least a small portion of the protein from a cloned DNA fragment. This method is, however, dependent on the use of a high-quality antibody directed against a desired protein. It does not matter whether the protein has been synthesized in vivo or in vitro. 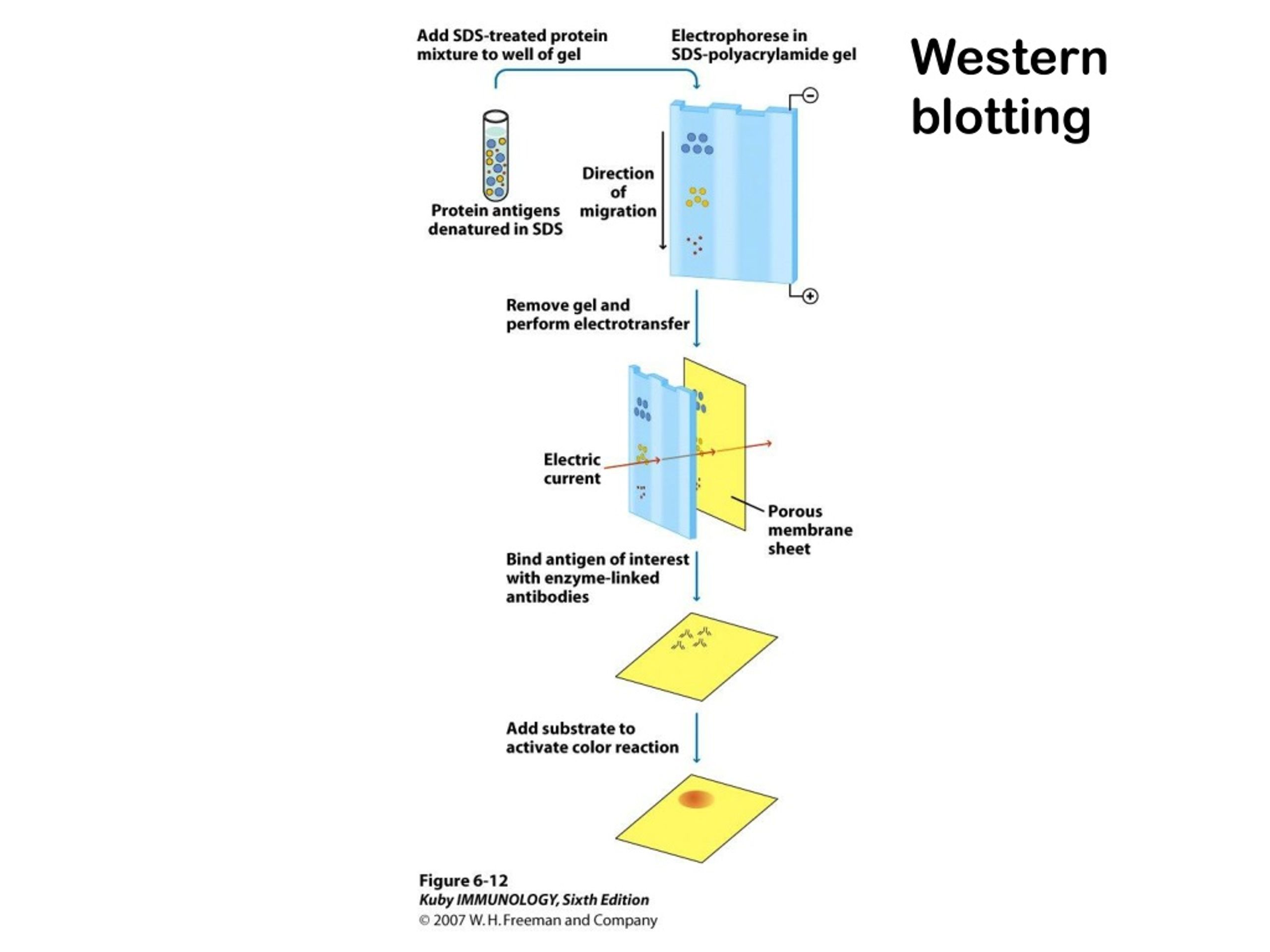
Western blot analysis can detect one protein in a mixture of any number of proteins while giving you information about the size of the protein.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
